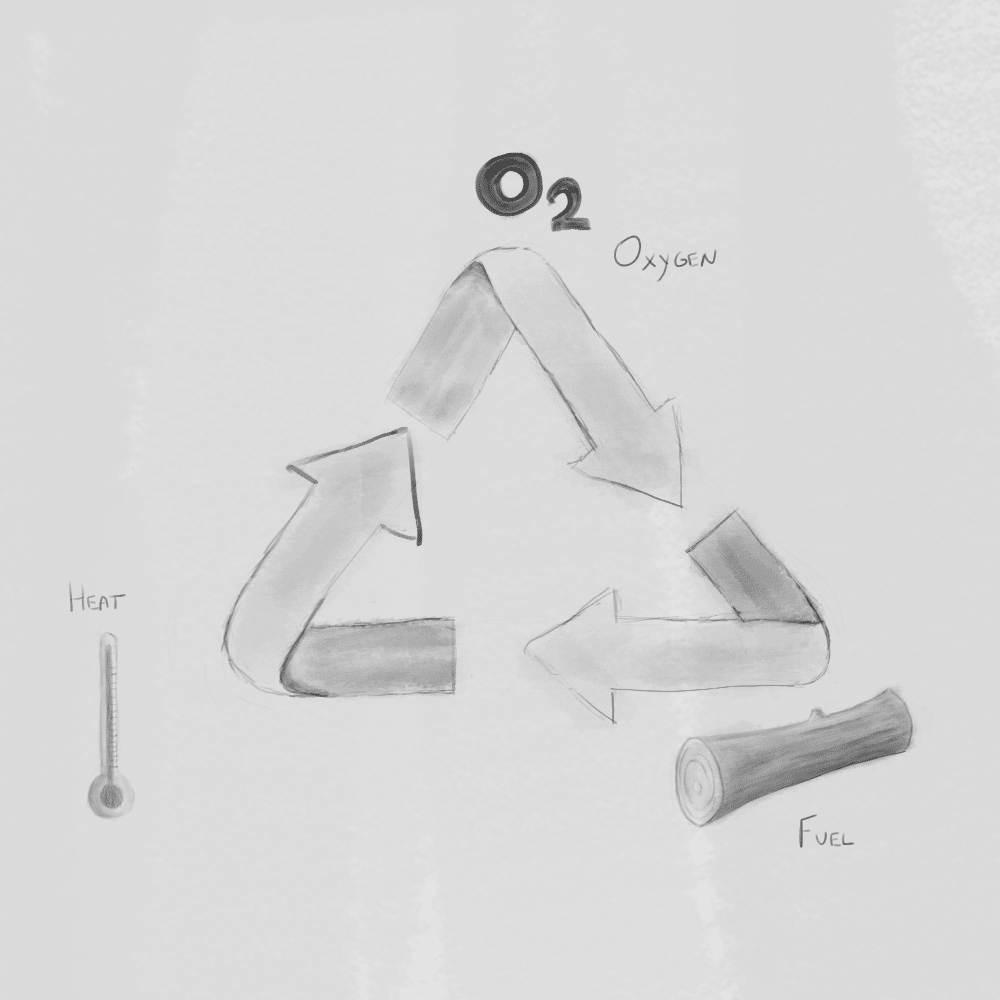A campfire is one of the most essential survival tools you can have in the outdoors. It provides warmth in cold conditions, allows you to cook food safely, boil water for purification, and even dry wet clothes after rain or river crossings. Beyond comfort, fire also plays a psychological role—boosting morale and giving you a sense of control in unfamiliar environments.
Even though we have many modern and convenient fire starters, our ancestors did not rely on them. Instead, they developed a wide range of techniques—many of which have been forgotten over time. These skills can be extremely valuable in unpredictable situations, where tools may fail or be unavailable. In this first part of the guide How to make a campfire – Science and Techniques, we’ll study what fire is and how it works.
What You’ll Learn in This Guide
In this first part, you’ll learn:
What is fuel and how does it burn?
What is fire?
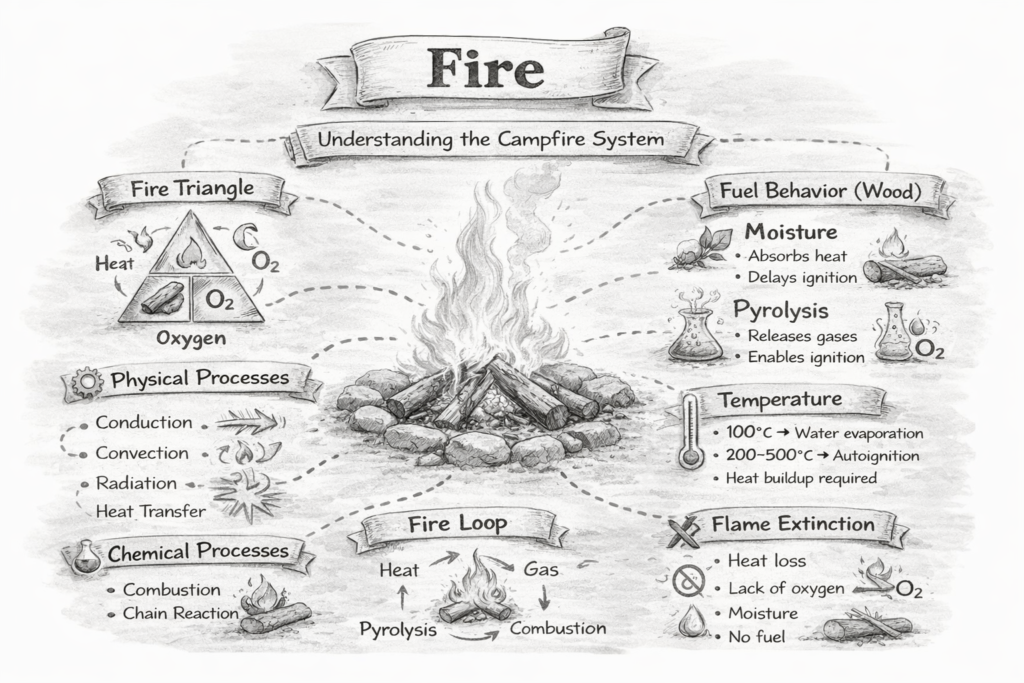
Fire is a rapid chemical reaction called combustion, where a fuel reacts with oxygen and releases energy in the form of heat and light. Although it looks simple, fire is actually a dynamic process driven by physics and chemistry working together.
For a fire to exist, three elements must be present—often called the fire triangle: fuel, oxygen, and heat. Fuel can be materials like wood, dry grass, or charcoal. Oxygen comes from the air, and heat is the initial energy needed to start the reaction. If any one of these elements is removed, the fire will weaken or go out completely.
One important concept to understand is that fire does not burn solid wood directly. When wood is heated, it undergoes a process called pyrolysis, where it breaks down and releases flammable gases. These gases are what actually ignite and produce the visible flame. In other words, when you see a flame, you are seeing burning gases, not the solid material itself.
Once the fire starts, it becomes a self-sustaining cycle. The heat from the flame continues to break down more fuel, releasing additional gases that keep feeding the fire. This cycle continues until the fuel is exhausted, oxygen becomes limited, or the temperature drops below what is needed to sustain combustion.
What is the fire triangle?
The fire triangle explains combustion as the interaction of three essential elements: heat, fuel, and oxygen. Heat raises the fuel to its ignition temperature, the fuel provides chemical energy, and oxygen supports the reaction. When these three elements are present in the right conditions, a fire can start and continue to burn.
However, this model is a simplification and does not fully describe what happens during combustion. It suggests that simply combining heat, fuel, and oxygen is enough, but in reality, fire involves more complex interactions. Physical processes like heat transfer and chemical changes inside the fuel play a critical role, especially during stages like drying and pyrolysis.
To better represent this, a fourth element is added: the chemical chain reaction. This transforms the triangle into the fire tetrahedron. The chain reaction sustains the fire by continuously generating heat and reactive particles, allowing combustion to persist as long as all four elements remain present.
This means fuel is not just a passive element in the fire triangle—it is an active system undergoing constant change. Its composition, moisture content, density, and structure all influence how easily it ignites, how intensely it burns, and how long the fire lasts.
Understanding fuel at this deeper level helps bridge the gap between the simplified fire triangle and the more complete fire tetrahedron, where combustion is seen as a dynamic and self-sustaining process.
Among these elements, fuel is often the least understood—especially when dealing with natural materials like wood.
What Is Fuel and How Does It Burn?
The term fuel refers to a substance that stores chemical potential energy, which can be released as thermal energy through a chemical reaction (combustion). However, fuel does not burn directly. Instead, it must first break down into flammable gases through heat.
The lowest temperature at which a fuel produces enough vapor to ignite under controlled conditions is called the flash point. Slightly above this is the fire point (or flame point), which is the temperature at which the fuel produces sufficient vapor to sustain a continuous flame.
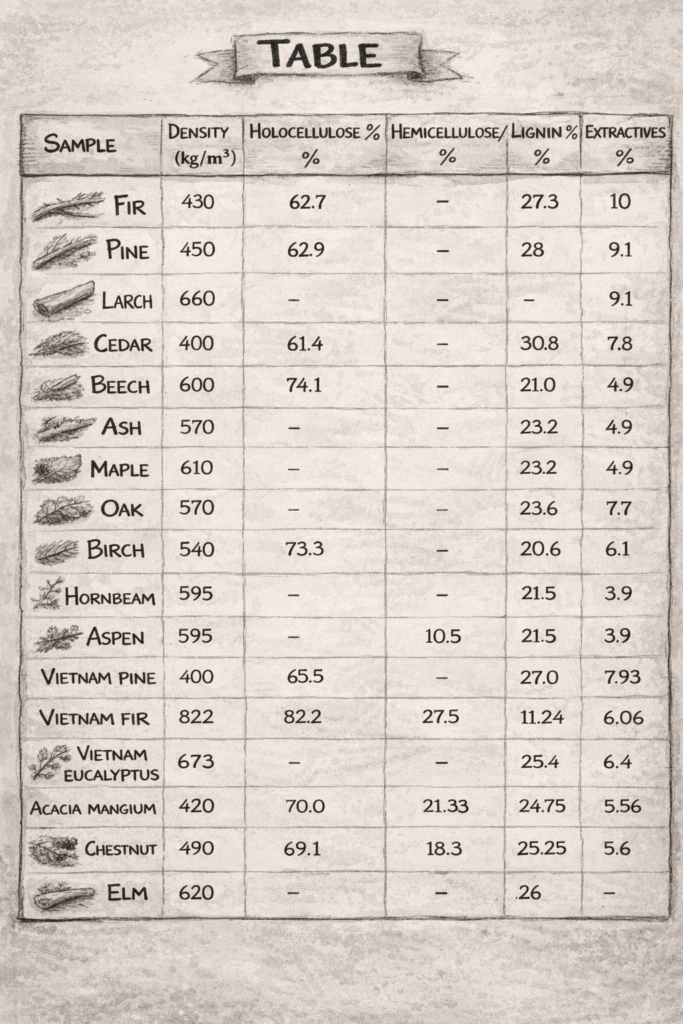
Different types of wood burn differently because each one has a unique internal composition, mainly consisting of:
Hemicellulose
The first component to decompose. It breaks down quickly into flammable gases, making ignition easier.
Decomposes at ~200–300°C
Cellulose
The main source of flammable gases. It sustains the flame once ignition begins.
Decomposes at ~300–400°C
Lignin
Produces more char (coal-like material). It is harder to ignite, but once ignited, it burns longer and contributes to stable embers.
Decomposes at ~250–500°C
Density
Density refers to how much mass is packed into a given volume of wood. Less dense woods contain more air spaces, allowing them to heat up and ignite more easily.
To distinguish between softwood and hardwood, keep in mind that:
What is hardwood and softwood?
Softwoods come from coniferous (cone-bearing), usually evergreen trees such as pine, spruce, cedar, and fir. They generally have lower density, grow faster, and are easier to work with.
Hardwoods come from angiosperm trees, which typically have broad leaves and produce seeds enclosed in fruits or nuts. They are usually denser and burn longer.
Understanding what fuel is gives us the foundation, but it still doesn’t explain how fire actually develops over time. Fuel is not consumed all at once—instead, it goes through a series of transformations as it is exposed to heat.
In materials like wood, combustion happens in distinct stages, each driven by physical and chemical changes inside the fuel. These stages determine how easily a fire starts, how it spreads, and how long it lasts.
This becomes especially important in friction-based fire methods (covered in Part 3 of this guide), where the goal is to generate enough heat to trigger pyrolysis and release flammable gases.
By looking at these stages, we move beyond the idea of fuel as a static element and begin to understand it as a dynamic process, constantly changing under the influence of heat and oxygen.
Stages of fire in wood
1. Drying (≈ 100°C)
- Moisture evaporates from the wood
- No combustion occurs yet, but heat is being absorbed (endothermic process)
2. Pyrolysis (≈ 200–350°C+)
Thermal decomposition of wood in limited or no oxygen. Wood breaks down into:
- Flammable gases (e.g., methane CH₄, carbon monoxide CO, hydrogen H₂, and other hydrocarbons)
- Liquid compounds (tars and oils)
- Solid residue (char/charcoal)
3. Ignition (≈ 300°C+, varies)
- Released gases mix with oxygen and ignite
- This is where visible flames begin
- Depends on fuel type, moisture, and oxygen availability
4. Combustion
Sustained burning process, including:
- Flaming combustion (gas-phase burning → visible flames)
- Smoldering combustion (surface oxidation of char → glowing embers, less flame)
5. Flame extinction
- Fire diminishes when one or more elements of the fire triangle (heat, oxygen, fuel) become insufficient
- Can leave behind unburned char or ash
- In practice, what we see as “burning wood” is actually the combustion of gases released during pyrolysis, not the solid wood itself.
Drying Stage
Remark
Specific heat: energy required to change a substance’s temperature
Latent heat: energy absorbed or released during a phase change (solid → liquid → gas) at constant temperature
The transition from pyrolysis to combustion is a delicate process. Many people think that the fire triangle consists only of three elements—fuel, oxygen, and heat—and, at a basic level, that is true. However, this simplified model does not fully describe what actually happens inside the material.
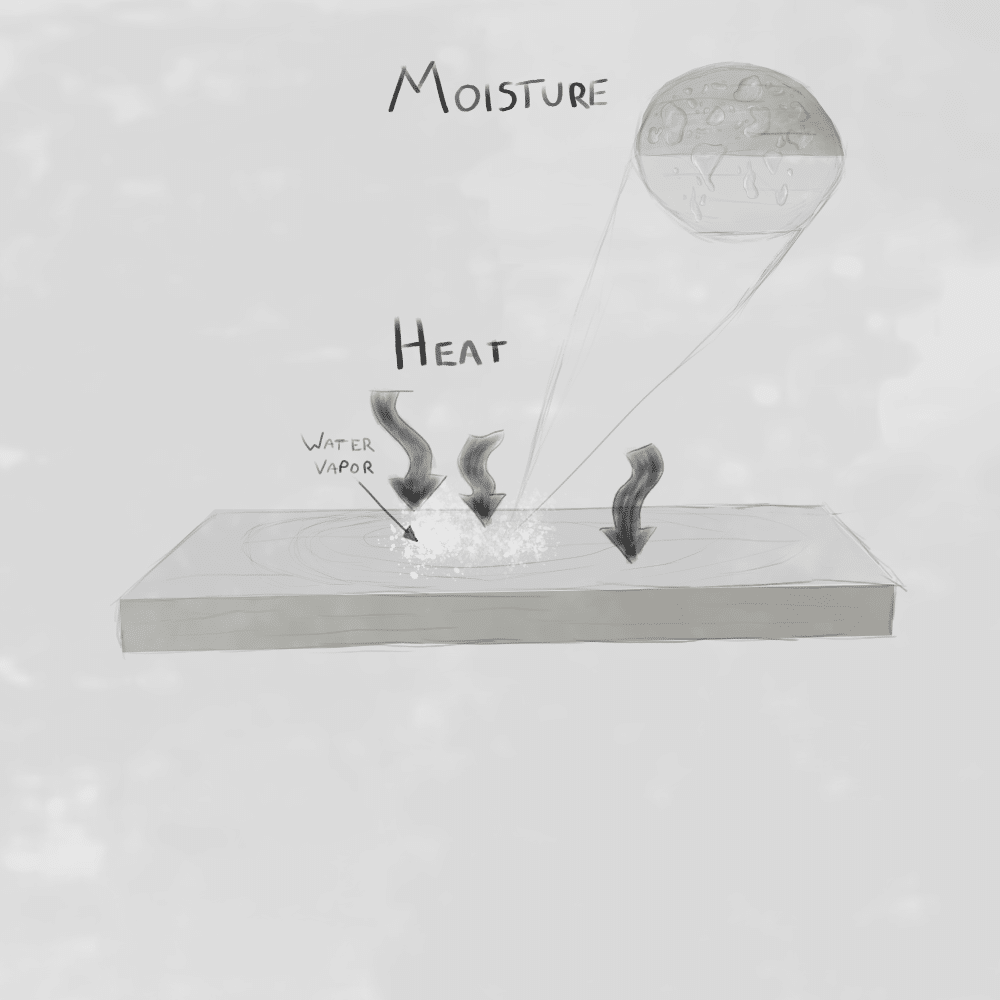
In reality, both physical and chemical processes begin as soon as the wood starts to dehydrate. The water molecules inside the wood absorb the energy that would otherwise heat the material.
Before wood can reach combustion temperatures (around 200–300°C+), a significant portion of the energy is first transferred to the water. This happens because water has a high specific heat capacity, meaning it can absorb a large amount of energy with only a small increase in temperature.
In addition, when water reaches its phase change temperature (around 100°C, depending on altitude), the temperature stops increasing. Instead, a lot of incoming energy is used for the phase transition from liquid to vapor. This is known as latent heat of vaporization. As a result, the energy that would normally drive pyrolysis is instead consumed by the evaporation process.
What happens physically inside the wood
Water fills the pores and cellular structure of the wood. As heat is applied:
- Water expands and turns into steam
- Steam escapes gradually from the material
- This creates a cooling effect and internal pressure, slowing down heating
What happens chemically
Combustion requires:
- Fuel (flammable gases released from wood)
- Oxygen
- Heat
Moisture interferes with all three:
Reduces temperature → prevents pyrolysis from occurring
Dilutes flammable gases → lowers combustion efficiency
Limits oxygen contact → disrupts the reaction
Pyrolysis Stage
What temperature starts a fire?
If you remember your physics classes, temperature is a measure of the average kinetic energy of particles within a system. All matter is composed of atoms, which combine to form molecules. These molecules are always in motion—even when the object appears still to us.
The lower the kinetic energy, the more rigid (solid) the material tends to be, although this varies depending on the substance. For example, a piece of steel can conduct heat much faster than wood (because of its higher thermal conductivity), but it is far more difficult to ignite. Wood, on the other hand, undergoes a process called pyrolysis, which makes combustion possible.
When wood is heated, it doesn’t burn immediately as a solid. Instead, it decomposes and releases flammable gases. Once these gases mix with oxygen and reach the right temperature, they ignite, producing heat and light in an exothermic oxidation reaction.
The temperature required for this ignition depends on the material and is known as the autoignition temperature. For most organic materials like wood, this typically occurs between 200°C and 500°C. At the lower end of this range, pyrolysis begins and small amounts of flammable gases are released. As the temperature increases, gas production intensifies until it reaches a point where ignition can occur without an external flame or spark.
This is why different materials ignite at different temperatures: their internal structure, density, and chemical composition determine how easily they decompose and release flammable gases. In practical terms, starting a fire means generating enough heat to push the material into this critical temperature range, where combustion becomes self-sustaining.
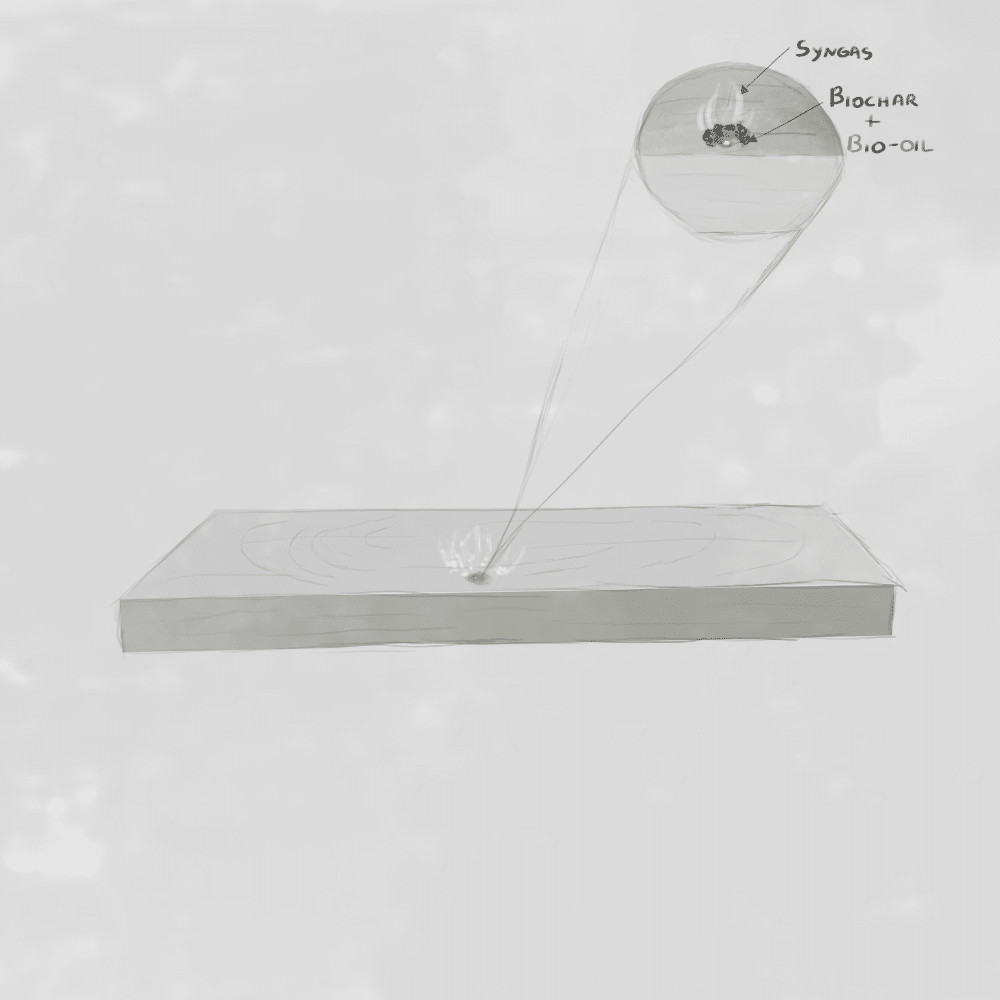
What is Pyrolysis?
Pyrolysis – a thermochemical decomposition of organic material at high temperatures in the absence of oxygen. This process breaks the material into three main products:
– Syngas (synthetic gas):
- Flammable gases such as CH₄, CO, H₂, and other hydrocarbons
- Non-flammable components like water vapor (H₂O), carbon dioxide (CO₂), and organic acids
– Liquid bio-oil and tars:
- Heavy vapors that condense into liquids. Includes methanol (historically called wood alcohol) and complex tar compounds such as phenols, furfural, and creosote.
– Solid biochar:
- A carbon-rich solid residue (charcoal)
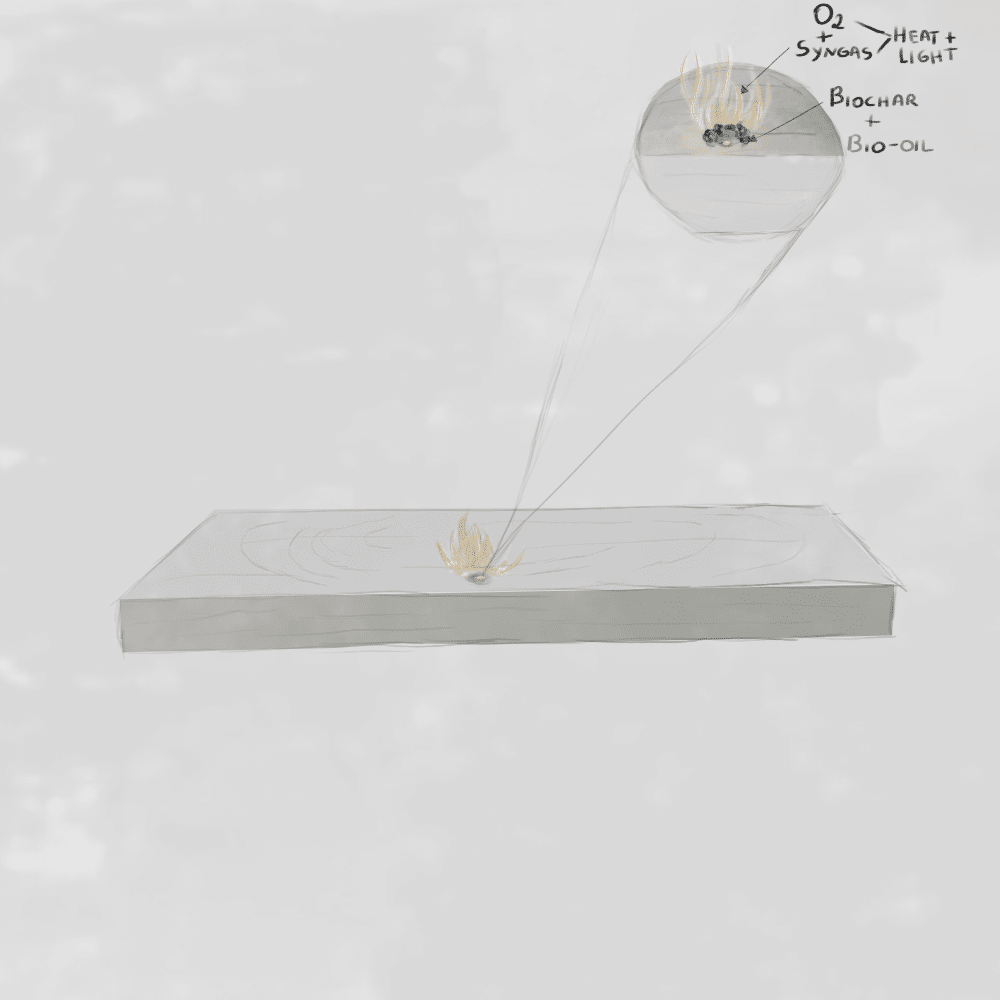
Once the flammable gases produced during pyrolysis mix with oxygen, combustion begins and a visible flame is formed.
When heating charcoal for a barbecue, people often use powerful or portable fans to accelerate the combustion process. Fanning the charcoal forces oxygen into the coals, significantly increasing the rate of combustion. This rapid influx of oxygen feeds the chemical reaction, producing more heat and causing the charcoal to burn hotter and faster.
Ignition and Combustion Stage
What is Autoignition?
Autoignition is the process by which a material ignites without an external flame or spark. It occurs when the material reaches a temperature high enough for its internal chemical reactions to begin spontaneously. At this point, the fuel releases flammable gases through pyrolysis, and these gases react with oxygen in the air, producing heat, light, and flame.
Ignition is the moment that starts the fire loop. Once the first gases ignite, they release heat that feeds back into the fuel. This heat is transferred through conduction, convection, radiation, and direct flame contact, raising the temperature of nearby material. As a result, more fuel undergoes pyrolysis, releasing additional gases that sustain and expand the reaction.
What is combustion?
Combustion is the chemical process that follows ignition. It is an exothermic oxidation reaction, where fuel reacts with oxygen to release energy in the form of heat and light. This process transforms solid fuel into gases, ash, and other byproducts. The visible flame is actually the combustion of these gases, not the solid wood itself.
The temperature at which combustion begins depends on the type of wood and its condition. Most woods autoignite between 200°C and 500°C, but factors such as density, moisture content, and chemical composition play a major role. Dry, softwoods tend to ignite more easily because they decompose faster and release gases more readily, while dense or moist hardwoods require higher temperatures and more sustained heat.
Once combustion begins, a self-sustaining cycle is created—often called the fire loop. Heat generated by the flame continues to dry the fuel, drive pyrolysis, and ignite new gases. As long as fuel, oxygen, and sufficient heat are present, this loop maintains the fire. Breaking any part of this cycle will cause the fire to weaken or extinguish.
Flame Extinguishing Stage
Flame extinction occurs when the fire can no longer sustain the chain of physical and chemical processes required for combustion. In simple terms, the fire goes out when one or more elements of the fire system—heat, fuel, oxygen, or the chemical reaction—are disrupted.
The most common cause is heat loss. If the temperature drops below the level required for pyrolysis, the fuel will stop releasing flammable gases. Without these gases, combustion cannot continue, and the flame disappears. This can happen naturally as the fuel is consumed or intentionally by cooling the fire, such as adding water.
External environmental conditions can significantly accelerate this process. In cold environments—such as snowy or rainy regions—low temperatures and high moisture levels affect both the air and the fuel. Water continuously absorbs a large portion of the energy generated by the fire and carries it away into the environment. As a result, the fire must produce more energy to raise the fuel to its ignition temperature, making it much harder to start and sustain.
A lack of oxygen also plays a key role. When airflow becomes restricted, the combustion reaction slows down and eventually stops. Covering a fire with soil, ash, or a fire blanket works because it blocks oxygen from reaching the fuel.
Fuel depletion or interruption of the reaction can also extinguish a flame. When the fire runs out of combustible material, or when moisture absorbs too much heat, the fire loop breaks. Without enough energy to sustain pyrolysis and combustion, the system collapses and the fire goes out.
In essence, extinguishing a fire means breaking the fire cycle. Whether by cooling, smothering, or removing fuel, the goal is always the same: to interrupt the balance that keeps the fire alive.
Conclusion
Fire is not a simple phenomenon, but a dynamic system where physical and chemical processes interact continuously. From the initial heating and dehydration of the fuel to pyrolysis, ignition, combustion, and eventual extinction, each stage depends on a precise balance of heat, oxygen, and fuel.
Understanding this process goes beyond theory—it gives you control. By recognizing how heat is generated, transferred, and lost, and how factors like moisture, airflow, and material properties influence combustion, you can better predict how a fire will behave in real conditions.
This knowledge is what separates guesswork from skill. Instead of simply trying to “make a fire,” you begin to manage a system—adjusting variables, responding to the environment, and maintaining the conditions needed for combustion.
With this foundation, you are now prepared to move from theory to practice.
What Comes Next
Now that you understand how fire works at a physical and chemical level, the next step is applying this knowledge in real outdoor conditions.
In Part 2, we’ll explore different campfire layouts and how they influence fire behavior, including airflow dynamics, wind direction, and structural stability. We’ll also cover practical strategies to maintain a fire in challenging environments, such as rain and snow, and how to protect your fire from moisture while keeping it efficient and sustainable.